Role of the Diffusion Layer in Energy Devices
© The Physical Society of Japan
This article is on
Convection Effect on Resistance Components in Liquid Thermoelectric Converter
(JPSJ Editors' Choice)
J. Phys. Soc. Jpn.
95,
044601
(2026)
.
In many energy devices, electron transfer occurs between ions dissolved in the electrolyte and the electrodes. For keep current flowing, new ions must be supplied continuously to the electrode surface from the bulk region. The diffusion layer’s role in diffusive mass transfer was clarified using the rotating disk electrode method.
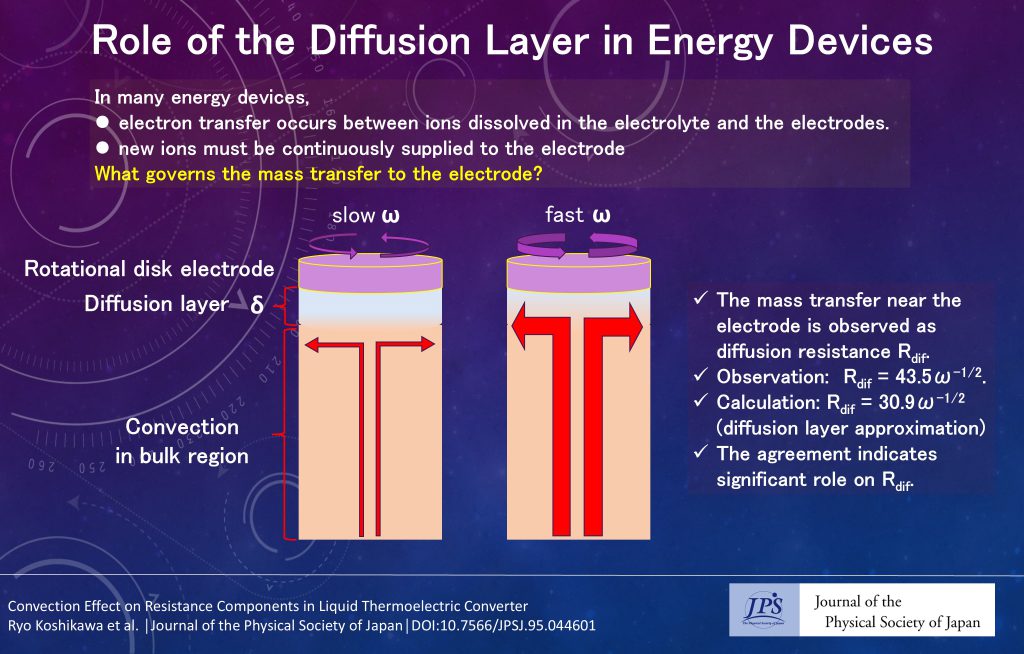
In energy devices, such as batteries and liquid thermoelectric converters, electron transfer occurs between ions dissolved in the electrolyte and the electrodes. For current to continue flowing in such devices, new ions must be supplied continuously to the electrode surface from the bulk region of the electrolyte. This is because ions that have undergone electron transfer cannot undergo further electron transfer. The driving force for mass transfer is the concentration difference ΔC between the electrode surface and the bulk region.
When an electric current I is passed through the electrolyte, ions are consumed at the electrode surface; hence, the ion concentration at the electrode surface decreases compared with the bulk region. The resulting concentration difference ΔC drives the diffusion of new ions to the electrode surface. Eventually, the concentration distribution near the electrode reaches a steady state. The concentration distribution at the steady state is usually approximated with a linear function (diffusion layer approximation), i.e., the concentration increases proportionally with distance from the electrode surface and becomes constant when it reaches the value of the bulk region. The distance δ at which the concentration reaches the bulk value is called the “thickness” of the diffusion layer. In the steady state, the current (= AFDΔC/δ, where A, F, and D are electrode area, Faraday constant, and diffusion constant, respectively.) caused by diffusive mass transfer matches I. The mass transfer is observed as diffusion resistance Rdif. Rdif can be considered the equilibrium potential shift resulting from ΔC divided by I. If ΔC/C (C is the concentration in the bulk region) is sufficiently small, Rdif becomes 2kBT/eI ×ΔC/C, where kB and T are the Boltzmann constant and temperature, respectively.
In this study, the relationship between the diffusive mass transfer and the diffusion layer was clarified using the rotating disk electrode method, in which centrifugal force causes the electrolyte near the electrode to flow outward, and convection occurs from the bulk region toward the electrode to compensate for this. With increasing angular velocity ω of the rotating electrode, the flow velocity toward the electrode increases. It was found that Rdif decreased sharply with increasing ω. Detailed analysis revealed that Rdif is proportional to ω-1/2 with a proportionality constant of 43.5 Ωs-1/2. In the diffusion layer approximation, δ in the rotational electrode method is proportional to ω-1/2. Then, Rdif (= 2kBTδ/eAFDC) is expected to be proportional to ω-1/2. From the parameters of the electrolyte and electrodes, the proportionality constant is calculated as 30.9 Ωs-1/2. The calculated value showed good agreement with the observed value, demonstrating that Rdif can be understood quantitatively in terms of the diffusion layer.
(Written by Yutaka Moritomo on behalf of all authors.)
Convection Effect on Resistance Components in Liquid Thermoelectric Converter
(JPSJ Editors' Choice)
J. Phys. Soc. Jpn.
95,
044601
(2026)
.
Share this topic
Fields
Related Articles
-
Unveiling the Nodal Topology of the Spin-Triplet Superconductor Candidate UTe2
Structure and mechanical and thermal properties in condensed matter
Superconductivity
2026-4-13
Using high-quality UTe2 (Tc = 2.1 K), we identify its nodal gap structure. Field-angle‑resolved specific‑heat reveals a b-axis singularity, supporting spin-triplet superconductivity with nodes along the b axis.
-
Toward Clarification of Physical Properties of Quasicrystals: Noncollinear Magnetic Orders in Icosahedral Approximants
Cross-disciplinary physics and related areas of science and technology
Electronic transport in condensed matter
Magnetic properties in condensed matter
2026-4-6
An effective model based on magnetic anisotropy arising from a crystalline electric field is constructed for icosahedral approximants, which not only explains measured ferromagnets and antiferromagnets but also reveals new types of noncollinear magnetic orders.
-
Creation of Chiral Phonons−How Lattice Chirality Imparts Angular Momentum to Phonons?
Structure and mechanical and thermal properties in condensed matter
2026-3-2
Chiral crystals host phonons with intrinsic angular momentum, whose quantization and energy splitting reflect the lattice chirality and reveal the microscopic features of interatomic interactions.
-
Topological Recast of Vortex Structures in Human Heart Blood Flow
Cross-disciplinary physics and related areas of science and technology
Electromagnetism, optics, acoustics, heat transfer, and classical and fluid mechanics
2026-2-16
We developed a new topological data analysis method to objectively identify the cardiac vortex structures. The method provides robust quantitative metrics for advancing cardiovascular diagnostics.
-
Rethinking Replica Analysis of Learning
Cross-disciplinary physics and related areas of science and technology
Statistical physics and thermodynamics
2026-1-19
The statistical physics analysis of learning parametric models was revisited by combining the replica method with a grand canonical ensemble and variational approach, enabling prediction error estimation for learning systems with real data.




