Descriptor of Thermogalvanic Cell for Waste-heat Harvesting
Department of Electrical Engineering and Bioscience, School of Advanced Science and Engineering, Waseda University
© The Physical Society of Japan
This article is on
Scaling Relation between Electrochemical Seebeck Coefficient for Fe2+/Fe3+ in Organic Solvent and Its Viscosity
(JPSJ Editors' Choice)
J. Phys. Soc. Jpn. 90, 033602 (2021).
This article is on
Waste Heat Harvesting: Descriptor of Thermogalvanic Cell
JPSJ News Comments 18, 07 (2021).
The scaling relation between the Seebeck coefficient and solvent viscosity would enable for the design of improved thermogalvanic cells.
Harvesting low-grade heat from industrial processes significantly improves energy efficiency in operating factory activities. Because converting thermal energy into electricity enables ceaseless access to clean energy, high-performance thermoelectric devices are an essential technology for sustainable development [1].
Thermogalvanic cells (or thermo-electrochemical cells), which are composed of a liquid electrolyte with hot and cold electrodes, are capable of efficiently converting thermal energy into electricity [2, 3]. However, a microscopic mechanism that dominates the performance of thermogalvanic cells is yet to be fully understood, hindering their performance improvement and widespread deployment.
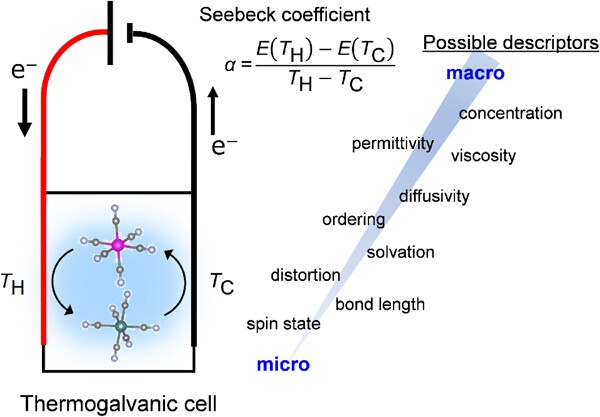
Fig. 1. Schematic illustration of a thermogalvanic cell. Determining the descriptors of the Seebeck coefficient of a redox couple is primarily important to achieve efficient waste-energy harvesting.
The research group led by Moritomo [4] identified the scaling relation between the Seebeck coefficient (α) of [FeIII(CN)6]3-/[FeII(CN)6]4- redox couple and the viscosity (η) of solvents, where α ranges from 0.14 mV/K (glycerin, η = 1412 mPa s) to 3.6 mV/K (acetone, η = 0.3 mPa s) as a function of η‒0.4. X-ray absorption near-edge structure (XANES) showed the deformation of Fe2+ coordination environment with decreasing η, suggesting that a low-η solvent weakly interacts with a solute to provide a large configuration-entropy change upon redox, and thus large α.
To clarify the origin for this scaling relation, the microscopic understandings of both η and α are required. Large-scale quantum mechanics/molecular mechanics (QM/MM) calculation of electrolytes could provide accurate electrolyte structures, shedding light on the atomistic mechanism [5]. In addition, considering a relatively low coefficient of determination (R2) for the scaling relation, α is also affected by other macroscopic/microscopic descriptors. Following systematic data collection, state-of-the-art machine learning analyses could determine discriminating features [6], and the resulting accurate performance prediction could enable the development of advanced thermogalvanic cells for sustainability.
References
[1] J. He and T. M. Tritt, Science 357, eaak9997 (2017).
[2] T. I. Quickenden and Y. Mua, J. Electrochem. Soc. 142, 3985 (1995).
[3] H. J. V. Tyrrell, D. A. Taylor, and C. M. Williams, Nature 177, 668 (1956).
[4] D. Inoue, H. Niwa, H. Nitani, and Y. Moritomo, J. Phys. Soc. Jpn. 90, 033602 (2021).
[5] Y. Tateyama, J. Blumberger, M. Sprik, and I. Tavernelli, J. Chem. Phys. 122, 234505 (2005).
[6] A. Chen, X. Zhang, and Z. Zhou, InfoMat 2, 553 (2020).
Scaling Relation between Electrochemical Seebeck Coefficient for Fe2+/Fe3+ in Organic Solvent and Its Viscosity
(JPSJ Editors' Choice)
J. Phys. Soc. Jpn. 90, 033602 (2021).
Waste Heat Harvesting: Descriptor of Thermogalvanic Cell
JPSJ News Comments 18, 07 (2021).

